Welcome to Krauß Lab
(Prof. Dr. Sybille Krauß)
Neurodegenerative diseases are characterized by the progressive loss of cells in the central nervous system. The loss of neurons leads to various symptoms, including dementia. An in-depth knowledge of the cellular mechanisms that contribute to the disease development is a prerequisite for the development of therapeutic approaches. While some neurodegenerative diseases occur sporadically, others are caused by genetic mutations.
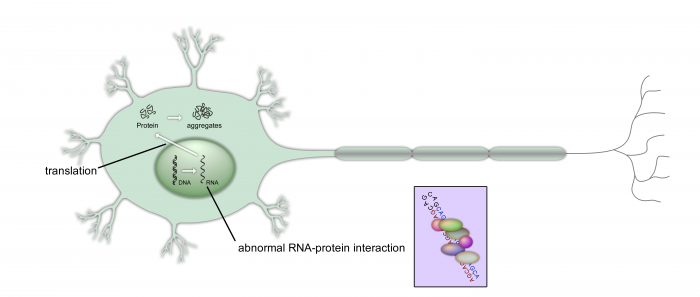
One of the most common causes of genetic neurodegeneration is expansion of short repetitive DNA sequences, for example CAG trinucleotide repeats. The so-called CAG repeat diseases are characterized by symptoms such as the progressive loss of cognitive abilities and progressive movement disorders. Although each of the CAG repeat expansion disorders is rare, together they represent one of the most common forms of inherited neurodegeneration. The CAG repeat can either be located in the coding region or in the untranslated region of the respective genes. When translated into protein the CAG repeat encodes for a polyglutamine stretch in the disease-causing proteins. In addition to encoding neurotoxic protein species, the CAG repeat RNA can have a toxic function itself. Structurally, mutant CAG repeat RNA differs from the physiological RNA: the expanded CAG repeats fold into characteristic hairpin structures. RNA-mediated toxicity can be explained by aberrant recruitment of RNA-binding protein to these hairpin structures.
The goal of our studies is to investigate such aberrant RNA-protein interactions and thereby identify molecular targets for the development of innovative treatment strategies. In our studies we have identified a set of proteins that aberrantly bind to mutant CAG repeats. This abnormal binding can lead to both: a loss of physiological function of the RNA-binding proteins and a gain of function of these proteins at the mutant CAG repeat RNA. For example, we have identified a protein complex, which binds to and induces aberrant translation of mRNAs containing mutant CAG repeats. Interestingly, the same protein complex promotes pathology in other diseases including sporadic neurodegenerative diseases, suggesting that deregulation of common cellular pathways may contribute to development of otherwise unrelated diseases. For our investigations we are using a broad panel of biochemical, molecular and cell biological methods.